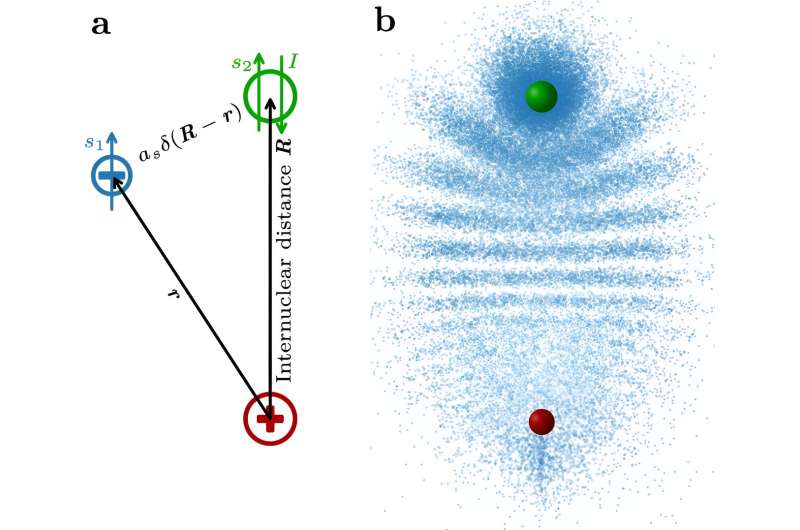
Sketch of a trilobite Rydberg molecule. A Sketch of a Rydberg molecule. The coordinates of Rydberg electrons (blue) and ground-state atoms (green) relative to the Rydberg core (red) are indicated by black arrows.The relevant spin is that of the Rydberg electron s1the electrons of the ground state atom s2 and the nuclear spin of the ground state atom I. Second Sketch of a trilobite molecule. Rydberg nuclei and ground-state atoms are shown as red and green spheres, respectively (size enlarged). The electron probability density projected into two dimensions is represented by the density of the blue dots. Credit: nature communications (2023). DOI: 10.1038/s41467-023-43818-7
Physicists from the doctoral team of Professor Herwig Ott in Kaiserslautern have succeeded for the first time in directly observing pure trilobite Rydberg molecules. What’s particularly interesting is that these molecules have very peculiar shapes that are reminiscent of trilobite fossils. They also have the largest electric dipole moment of any molecule known to date.
The researchers used a specialized device that allows them to prepare these fragile molecules at ultra-low temperatures. The results revealed their chemical binding mechanism, which is different from all other chemical bonds.The research is published in the journal nature communications.
In their experiments, the physicists used a cloud of rubidium atoms cooled in ultrahigh vacuum to about 100 microkelvin, 0.0001 degrees above absolute zero. They then used lasers to excite some of these atoms into what’s known as the Rydberg state. “In this process, the outermost electrons are in each case brought into distant orbits around the atomic body,” says Herwig, who studies ultracold quantum gases and quantum atomic optics at the University of Kaiserslautern-Landau. ·Professor Ott explains.
“Electrons can have orbital radii exceeding one micron, making electron clouds larger than small bacteria.” Such highly excited atoms also form in interstellar space and are extremely chemically reactive.
If the ground state atoms were now inside this giant Rydberg atom, a molecule would form. While standard chemical bonds are covalent, ionic, metallic or dipolar, trilobite molecules are held together by an entirely different mechanism.
“Quantum mechanical scattering of the Rydberg electrons in the ground-state atom sticks the two together,” said Max Althn, first author of the study. “Imagine the electrons spinning rapidly around the nucleus. On each round trip , it collides with the ground-state atom. Contrary to our intuition, quantum mechanics tells us that these collisions result in an effective attraction between the electron and the ground-state atom. “
The properties of these molecules are stunning: Due to the wave nature of electrons, multiple collisions create interference patterns that look like trilobites. Furthermore, the molecule’s bond lengths are as large as Rydberg orbitals, much larger than those of other diatomic molecules. Because electrons are strongly attracted to ground-state atoms, the permanent electric dipole moment is extremely large: over 1,700 Debye.
To observe these molecules, scientists developed specialized vacuum devices. It allows the preparation of ultracold atoms through laser cooling and subsequent spectroscopic detection of the molecules. These results help us understand the fundamental binding mechanisms between ground-state atoms and Rydberg atoms, which have recently emerged as a promising platform for quantum computing applications. The researchers’ findings add to the understanding of the Rydberg system, which is both strange and useful.
More information:
Max Althn et al., Exploring the vibrational series of pure trilobite Rydberg molecules, nature communications (2023). DOI: 10.1038/s41467-023-43818-7
Provided by TU Kaiserslautern-Landau Rhein-Palatinate
citation: Research: Physicists create giant trilobite Rydberg molecule (2023, December 18), Retrieved December 23, 2023, from https://phys.org/news/2023-12-physicalists- giant-trilobite-rydberg-molecules.html
This document is protected by copyright. No part may be reproduced without written permission except in the interests of fair dealing for private study or research purposes. Content is for reference only.
#Study #Physicists #create #giant #trilobite #Rydberg #molecules
Image Source : phys.org